
Digital Endpoints for Clinical Trials
Real-time monitoring of medication efficacy and side effects.

Features Grid
Digital endpoints and real-time monitoring for pharmaceutical clinical trials.
Digital Endpoints
Validated measures of depression & anxiety.
Rapid Non-Response Detection
Within 4–6 days.
Post-Marketing Surveillance
Real-world evidence generation.
Evidence Section
Olanzapine and Lithium outcome data collected via Psynary supported research into early response patterns.
Medication Effectiveness Tracking
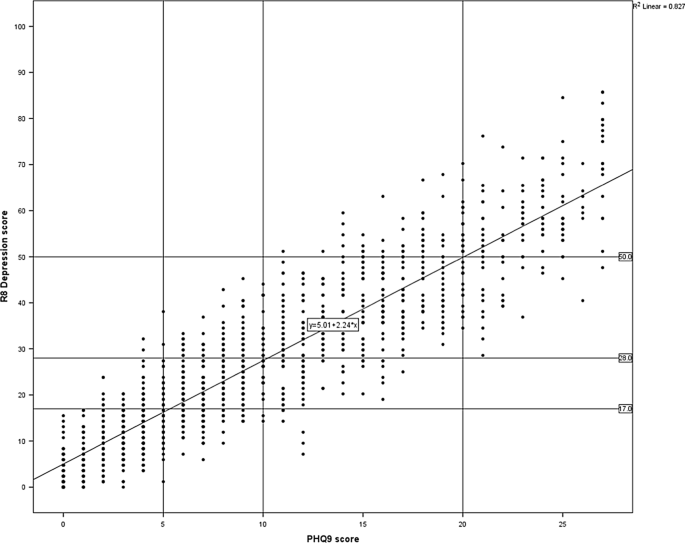
Benefits
Reduced Trial Costs
Faster recruitment and early efficacy detection reduce overall costs.
Higher-Quality Regulatory Submissions
Digital endpoints provide robust data for regulatory approval.
Continuous Monitoring Beyond Phase IV
Post-marketing surveillance and real-world evidence generation.
Accelerate Drug Development
Collect structured patient-reported data to support trial timelines, data quality, and researcher review with Psynary's digital data collection platform.
Integrate Psynary into your next trial
Partner with Psynary to collect structured patient-reported data for clinical trials and post-marketing studies.